What is photoelectron spectroscopy?
PES is an important analytical technique because it exposes a flaw in the Bohr model of the atom. Just as line emission spectra refined the nuclear model of the atom presented by Rutherford, PES data refines the quantized orbit model presented by Bohr. PES data suggests the existence of subshells corroborating the quantum mechanical model of the atom.
Background
-When light with sufficient energy interacts with atoms on the surface of a solid element, the electrons can be ejected. Recall that the energy of a photon is related to the wavelength () and frequency () of the photon. The equations are shown below:
E=hυ E=hc
*h is planck's constant*
-Photoelectron spectroscopy starts with the photoelectric effect but then uses an analyzer to measure
the kinetic energy of the ejected electrons along with the relative number of electrons having the same
kinetic energy.
-The kinetic energy of the ejected electrons can be measured, usually in electron volts
(1 eV = 1.60 x 10^-19 J). Using the law of conservation of energy, the measured kinetic energy can be
compared to the energy of the initial light leading to the binding energy (BE) of the electrons.
-The binding energy is an approximation of how much energy is required to eject an electron. The
expression for this relationship is shown below.
BE=hυphoton-KE
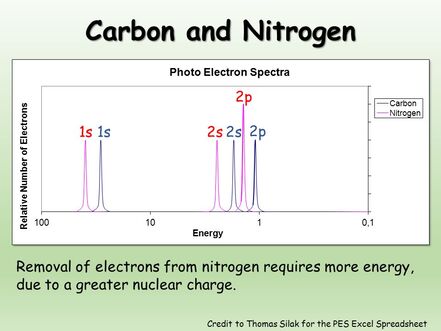
Therefore, the measured kinetic energy can be converted to the binding energy of the electrons leading to a spectrum like the one shown below. Electrons having higher binding energy experience a greater attraction to the nucleus. The peaks can be identified using Coulomb’s law to qualitatively rank the attraction of the electrons in an atom to the nucleus.
E=hυ E=hc
*h is planck's constant*
-Photoelectron spectroscopy starts with the photoelectric effect but then uses an analyzer to measure
the kinetic energy of the ejected electrons along with the relative number of electrons having the same
kinetic energy.
-The kinetic energy of the ejected electrons can be measured, usually in electron volts
(1 eV = 1.60 x 10^-19 J). Using the law of conservation of energy, the measured kinetic energy can be
compared to the energy of the initial light leading to the binding energy (BE) of the electrons.
-The binding energy is an approximation of how much energy is required to eject an electron. The
expression for this relationship is shown below.
BE=hυphoton-KE
Therefore, the measured kinetic energy can be converted to the binding energy of the electrons leading to a spectrum like the one shown below. Electrons having higher binding energy experience a greater attraction to the nucleus. The peaks can be identified using Coulomb’s law to qualitatively rank the attraction of the electrons in an atom to the nucleus.
|
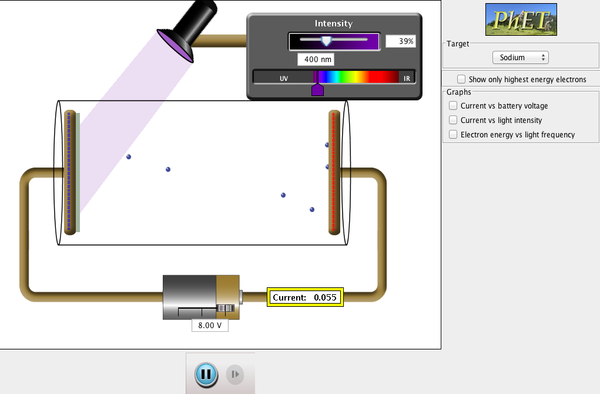
Introductory Questions (Analyze): Use the following link if you need help or want more information. You may also use your notes from class to aid in answering. https://www.khanacademy.org/science/chemistry/electronic-structure-of-atoms/bohr-model-hydrogen/a/photoelectric-effect 1. Define threshold frequency. Note that this frequency will correspond to a wavelength and energy. 2. What is the relationship between photon frequency and the kinetic energy of the ejected electrons? Phet simulation of the photoelectric effect: https://phet.colorado.edu/en/simulation/legacy/photoelectric Focus on how changing the wavelength affects the simulation. Once the threshold energy is reached, many different electrons are simultaneously ejected from the atoms. Because of the battery, the electrons are ejected and then replaced so the sample of atoms stays neutral. **This is different from ionization energy where one electron is removed per input of energy and second, third, fourth, etc. electrons are removed from a cation.** 3. What changes increase the measured current in the simulation? https://www.khanacademy.org/science/chemistry/electronic-structure-of-atoms/electron-configurations-jay-sal/a/photoelectron-spectroscopy 4. Are binding energy and kinetic energy of photoelectrons inversely or directly proportional? 5. What are the two types of photons generally used in PES? Follow-Up Questions (Apply): Draw a diagram of the classic Bohr model for a carbon atom. Write the electron configuration for a carbon atom. Use the PES spectrum of carbon provided in the Introduction to answer the following questions.
Provide the shell and subshell of the electrons having the highest binding energy & highest kinetic energy. Explain how this spectrum would look different if the Bohr model of the atom were accurate for all elements The actual binding energy for a 2p electron in carbon is 11.26 eV.
Conclusion: In your own words, summarize photoelectron spectroscopy and briefly explain how to use a PES spectrum to identify an element. |
Move That Magnet!
Background
In this inquiry lab, you will explore with magnets to see and feel attractive and repulsive forces. You will develop your own lab tests to identify the variables that affect forces of attraction or repulsion – the foundation of Coulomb’s Law.
Problem
How can you make the magnet move the most without touching it?
Procedure
*Write observations and any notes you may have*
Analysis Questions
Conclusion
In your own words, summarize variables that affect forces of attraction or repulsion.
In this inquiry lab, you will explore with magnets to see and feel attractive and repulsive forces. You will develop your own lab tests to identify the variables that affect forces of attraction or repulsion – the foundation of Coulomb’s Law.
Problem
How can you make the magnet move the most without touching it?
Procedure
- Obtain two ceramic magnets. Hold the magnets close to one another. Flip one of the magnets over and repeat. Based on the attractive or repulsive forces you feel, label each side of the magnet as “+” or “-.” Use a small piece of tape and marker to label each side of the magnet with “+” for positive and/or “-“ for negative. Repeat with all the other magnets, including neodymium magnets. Record your observations in the “Observation and Notes” section.
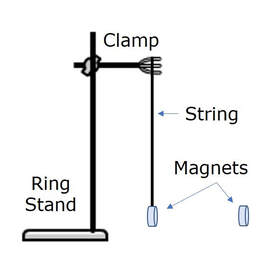
- Attach a clamp to a ring stand as shown at right.
- Tape a magnet to one end of a string. Tie the other end of the string to the clamp as shown at right. The magnet should be suspended in air and should not touch the table.
- Using this set-up, ruler, and additional ceramic and neodymium magnets, determine how to make the suspended magnet move the farthest (in cm). You may not touch the magnet. You may not use wind/air to move the magnet.
*Write observations and any notes you may have*
Analysis Questions
- How did you know which side of the magnet was positive and which side was negative? Explain using evidence and reasoning from your observations of the magnets.
- How far, in centimeters, were you able to move the suspended magnet?
- How did you make the magnet move the most without touching it? What method(s) worked best? Did other lab groups in the class see similar results?
Conclusion
In your own words, summarize variables that affect forces of attraction or repulsion.